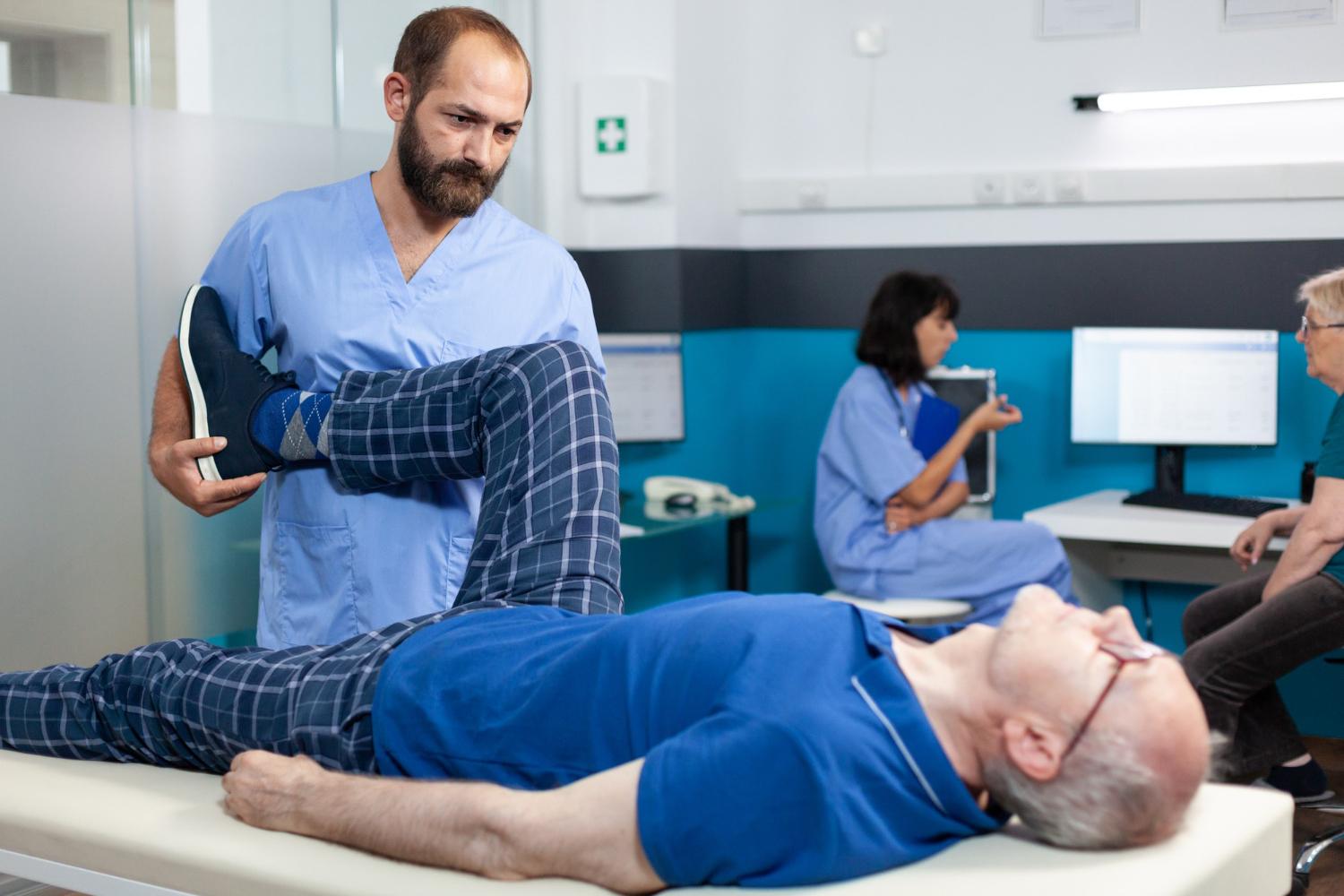
Comprehensive Chiropractic Care at a Modern Calgary Chiropractic Clinic
A modern Calgary chiropractic clinic focuses on delivering patient-centred care that supports long-term #spinal_health and overall wellness. By combining evidence-based techniques with up-to-date clinical knowledge, chiropractors aim to address the root causes of discomfort rather than only temporary symptoms. This approach aligns with current healthcare standards and emphasizes safety, #transparency, and professional expertise.
Working with an experienced Calgary chiropractor allows individuals to receive personalized treatment plans based on their #health_history, posture, lifestyle, and specific concerns ( https://maps.app.goo.gl/ehcGdSnte7SVFPw46 ). Services often include spinal adjustments, soft tissue techniques, posture correction strategies, and rehabilitative exercises. These methods support mobility, #reduce_tension, and promote proper nervous system function, which is essential for everyday activities.
Many households benefit from the care provided by a qualified family chiropractor, as treatment options are adapted for adults, seniors, and children. From managing work-related strain to #supporting_active_lifestyles, chiropractic care is tailored to meet changing needs at every stage of life. A focus on education helps patients understand their condition and participate actively in their recovery process.
Professional chiropractic in Calgary follows regulated clinical standards and ethical practices. Clinics prioritize clear communication, #informed_consent, and ongoing assessment to ensure appropriate care. A skilled Calgary chiro integrates manual therapy with ergonomic guidance and #wellness_recommendations, creating a balanced approach to musculoskeletal health.
Comprehensive chiropractic care is not limited to pain management alone ( https://myolab.ca/chiropractic/ ). It also supports injury prevention, improved posture, and functional movement. Choosing a clinic that values experience, continuing education, and patient-focused outcomes helps ensure care that is #reliable, responsible, and aligned with modern healthcare expectations.
Explore Our Website : https://paulasplate.net/discover-the-best-chiropractic-for-optimal-health/
A modern Calgary chiropractic clinic focuses on delivering patient-centred care that supports long-term #spinal_health and overall wellness. By combining evidence-based techniques with up-to-date clinical knowledge, chiropractors aim to address the root causes of discomfort rather than only temporary symptoms. This approach aligns with current healthcare standards and emphasizes safety, #transparency, and professional expertise.
Working with an experienced Calgary chiropractor allows individuals to receive personalized treatment plans based on their #health_history, posture, lifestyle, and specific concerns ( https://maps.app.goo.gl/ehcGdSnte7SVFPw46 ). Services often include spinal adjustments, soft tissue techniques, posture correction strategies, and rehabilitative exercises. These methods support mobility, #reduce_tension, and promote proper nervous system function, which is essential for everyday activities.
Many households benefit from the care provided by a qualified family chiropractor, as treatment options are adapted for adults, seniors, and children. From managing work-related strain to #supporting_active_lifestyles, chiropractic care is tailored to meet changing needs at every stage of life. A focus on education helps patients understand their condition and participate actively in their recovery process.
Professional chiropractic in Calgary follows regulated clinical standards and ethical practices. Clinics prioritize clear communication, #informed_consent, and ongoing assessment to ensure appropriate care. A skilled Calgary chiro integrates manual therapy with ergonomic guidance and #wellness_recommendations, creating a balanced approach to musculoskeletal health.
Comprehensive chiropractic care is not limited to pain management alone ( https://myolab.ca/chiropractic/ ). It also supports injury prevention, improved posture, and functional movement. Choosing a clinic that values experience, continuing education, and patient-focused outcomes helps ensure care that is #reliable, responsible, and aligned with modern healthcare expectations.
Explore Our Website : https://paulasplate.net/discover-the-best-chiropractic-for-optimal-health/
Comprehensive Chiropractic Care at a Modern Calgary Chiropractic Clinic
A modern Calgary chiropractic clinic focuses on delivering patient-centred care that supports long-term #spinal_health and overall wellness. By combining evidence-based techniques with up-to-date clinical knowledge, chiropractors aim to address the root causes of discomfort rather than only temporary symptoms. This approach aligns with current healthcare standards and emphasizes safety, #transparency, and professional expertise.
Working with an experienced Calgary chiropractor allows individuals to receive personalized treatment plans based on their #health_history, posture, lifestyle, and specific concerns ( https://maps.app.goo.gl/ehcGdSnte7SVFPw46 ). Services often include spinal adjustments, soft tissue techniques, posture correction strategies, and rehabilitative exercises. These methods support mobility, #reduce_tension, and promote proper nervous system function, which is essential for everyday activities.
Many households benefit from the care provided by a qualified family chiropractor, as treatment options are adapted for adults, seniors, and children. From managing work-related strain to #supporting_active_lifestyles, chiropractic care is tailored to meet changing needs at every stage of life. A focus on education helps patients understand their condition and participate actively in their recovery process.
Professional chiropractic in Calgary follows regulated clinical standards and ethical practices. Clinics prioritize clear communication, #informed_consent, and ongoing assessment to ensure appropriate care. A skilled Calgary chiro integrates manual therapy with ergonomic guidance and #wellness_recommendations, creating a balanced approach to musculoskeletal health.
Comprehensive chiropractic care is not limited to pain management alone ( https://myolab.ca/chiropractic/ ). It also supports injury prevention, improved posture, and functional movement. Choosing a clinic that values experience, continuing education, and patient-focused outcomes helps ensure care that is #reliable, responsible, and aligned with modern healthcare expectations.
Explore Our Website : https://paulasplate.net/discover-the-best-chiropractic-for-optimal-health/
0 Comments
0 Shares